Content Status
Type
Linked Node
Second Line LPA [SLLPA]
Learning ObjectivesProvide an overview of the SLPA
Indications for use of SLLPA
Results it provides and its interpretations
H5Content
Content
Second-line Line Probe Assay (SL-LPA) is a LPA to detect resistance to the second-line anti-TB drugs. This test is recommended for identifying TB patients with Multidrug-resistance (MDR) or rifampicin-resistance and those who can be placed on the shorter MDR-TB regimen.
The SL-LPA produces results in just 24-48 hours.
- It is performed using GenoType MTBDRsl Version 2.
- This is a DNA-based test that identifies genetic mutations in MDR-TB strains, making them resistant to fluoroquinolones and injectable second-line TB drugs.
- GenoType MTBDRsl Version 2 (Figure) identifies:
- Quinolone-resistance determining region (QRDR) of gyrA (from codon 85 to 96) and of gyrB (from codon 536 to 541) genes for detection of resistance to fluoroquinolones
- rrs (nucleic acid position 1401, 1402 and 1484), eis promoter region (from -37
to -2 nucleotides upstream, low level kanamycin resistance) for detection of resistance to SLI drugs.
- Single mutation in gyrA confers low level fluoroquinolone resistance.
- Concurrent mutations in gyrA or both gyrA and gyrB confer high level fluoroquinolone resistance.
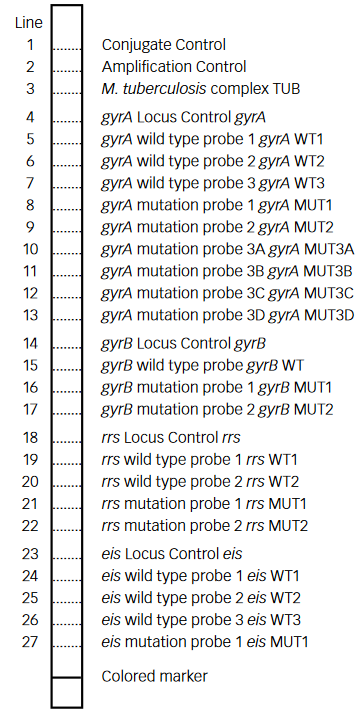
Figure: GenoType MTBDRsl Version 2 SL-LPA strip to determine Wild Type genes and mutations responsible for Resistance to Fluoroquinolones and Second-line Injectable Drugs; Source: GLI LPA Guidance Document.
Resources
Kindly provide your valuable feedback on the page to the link provided HERE
Page Tags
LMS Page Link
Content Creator
Reviewer
- Log in to post comments
Comments
Approved in B7
Namrata Tue, 22/02/2022 - 17:20
Approved in B7